When we discuss skin ageing, there’s one topic that must always be front and centre: UV damage. With sun damage shown to be responsible for 80 per cent of skin ageing, its contribution and strategies to minimise its impacts are highly important to protecting the lifespan of our skin.
Cumulative UV exposure
As UV hits the skin, DNA is altered and cellular repair kicks into action. Our bodies are able to repair some of this damage, but as we age and the more sun exposure we get, the less able our body is to fi x this damage. Repeated exposure means our bodies repair more slowly and less effectively, leading to an accumulation of damage that goes unrepaired. Subsequently, this can increase mutation burden, accelerate photoageing and increase cancer risk.
Genomic instability
UV radiation is the dominant source of DNA damage in skin and the major contributor to genomic instability. Stable genomes are able to repair damage effectively, maintain replication integrity and preserve chromosome structure. Conversely, unstable genomes accumulate mutations more quickly, cause impaired DNA repair and may develop chromosome abnormalities.
That means, when we talk about cumulative UV exposure, what we’re really talking about at a biological level is genomic instability, one of the most recognised hallmarks of ageing.
Genomic sequencing studies have shown that clinically normal-looking, sun-exposed skin carries an extremely high burden of somatic mutations, comparable to those seen in some solid tumours, yet still maintains function and integrity.
Persistent DNA damage
This apparent paradox refl ects the effectiveness of several compensatory systems acting together, including DNA damage response and repair pathways, regulated keratinocyte turnover and immune surveillance. However, persistent DNA damage acts as an upstream driver of ageing.
As repair capacity gradually declines, DNA lesions accumulate, promoting cellular senescence, chronic low-grade infl ammation and impaired regeneration. With age and ongoing cumulative UV exposure, skin shifts from effective compensation towards cumulative photodamage and, if unchecked, field cancerisation.
Photoprotection as genome protection
When we discuss it in this context, sunscreen then becomes genome protection, not simply photoprotection.
Sunscreen reduces the rate of UV-induced DNA lesion formation and fewer lesions mean lower genomic instability. This helps preserve normal cellular function and immune regulation over time.
Limiting environmentally driven DNA damage is foundational to maintaining skin function across the skin’s lifespan. Reducing UV exposure to the skin is the most direct way we can infl uence that biology.
Where do DNA repair enzymes fit in?
It’s important to fi rst note that DNA repair enzymes do not replace sunscreen. When appropriately delivered (through targeted systems such as liposomal technology), they can help accelerate repair of UV-induced DNA lesions that still occur. Clinical evidence is strongest in heavily sun-exposed, high-risk skin, where enhanced repair reduces molecular markers of photodamage.
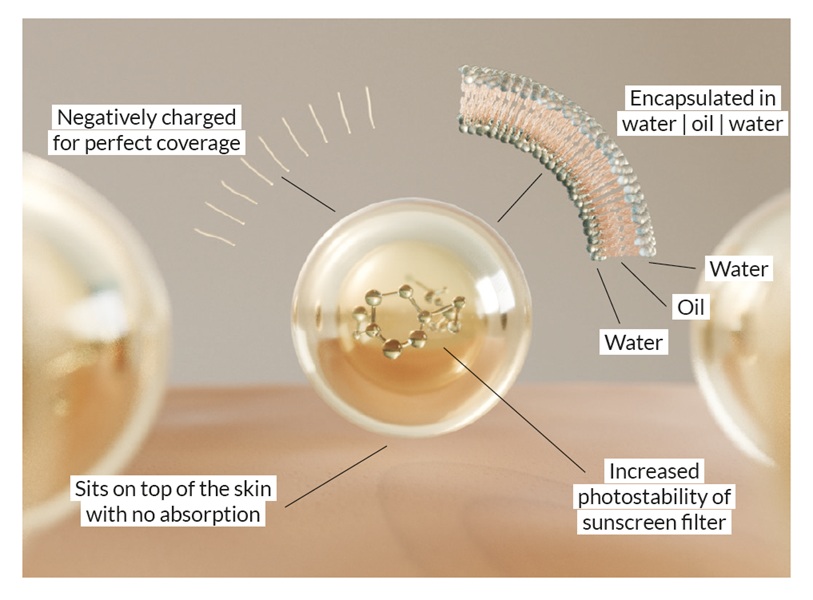
Together, sunscreen reduces damage and DNA repair determines how long skin can compensate before dysfunction emerges.
Next-generation protection skincare

The Ultimate Skin Ageing duo is designed as a one-step repair system that protects from future damage and repairs past damage through encapsulated sunscreen fi lters and two types of DNA repair enzymes.
Photolyase: An enzyme we need to help our body repair DNA, but an ability we lost through evolution. Working to fi nd and repair cyclobutane pyrimidine dimers (CPDs), small chinks in our DNA helix, by using a photon of light to give it energy and repair the CPD damage instantly through a process called photoreactivation.
UV Endonuclease: Our body repairs DNA through nucleotide excision repair (NER), however, as we age, this repair slows. UV Endonuclease works to enhance our natural NER and does not require a photon of light to work.
References: Flament et al., J Invest Dermatol (2013) https://pmc.ncbi.nlm.nih.gov/articles/PMC3790843/. Furman et al., Nature Aging (2025) https://pubmed.ncbi.nlm.nih.gov/40527938/. Schumacher et al., Nature (2021): https://doi.org/10.1038/s41586-021-03307-7. Martincorena et al., Science (2015): https://doi.org/10.1126/science.aaa6806. Wolf et al., J Invest Dermatol (2000): https://pubmed.ncbi.nlm.nih.gov/10620131/. Yarosh et al., Lancet (2001): https://pubmed.ncbi.nlm.nih.gov/11289350

 Added to basket
Added to basket

 Unapplied Changes
Unapplied Changes