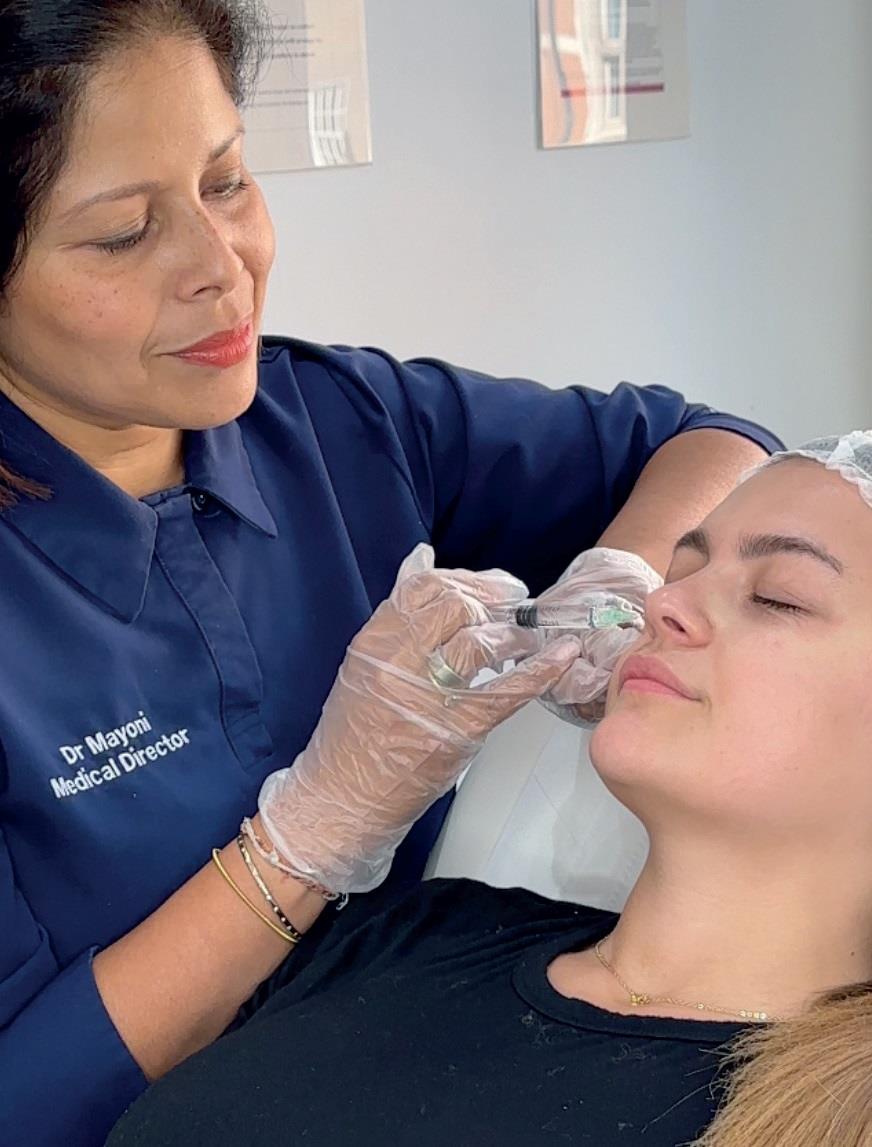
The Department of Health and Social Care recently released its consultation document on the future of licensing for aesthetic treatments in England. The open consultation considers which treatments could be included in a license for practitioners, and invites responses from the public.
Back in April 2022, the Health and Care Act gave the Secretary of State for Health and Social Care the power to introduce a licensing regime for non-surgical cosmetic procedures in England. The purpose of the scheme is to ensure that consumers undergoing a non-surgical cosmetic procedure can be confident that the treatment they receive is safe and of a high standard.
Any licensing scheme for non-surgical cosmetic procedures must strike a balance between protecting the public and building confidence in the safety of the aesthetic industry, while respecting consumer choice and encouraging innovation.
As we know, all procedures have some risks and can lead to complications if not correctly performed. These risks are greater where the person carrying out the procedure is not sufficiently knowledgeable or trained, where they use unregulated products, or when the procedure is carried out at unsuitable premises.
The current regulatory framework places few restrictions on who can perform non-surgical cosmetic procedures. The government recognises the concerns about the lack of regulation in this field and the potential dangers to the public. A regulatory framework will enable consumers to make informed and safe choices when undergoing procedures which have the potential to cause serious injury or harm.
The scheme will ensure that those who offer specified procedures:
• are suitably knowledgeable, trained and qualified
• hold appropriate indemnity cover
• operate from premises which meet the necessary standards of hygiene, infection control and cleanliness
Under the proposed scheme, which will be operated by local authorities in England, practitioners will need to be licensed to perform specific non-surgical cosmetic procedures and their premises will also need to be licensed.
At Wigmore Medical we believe that public safety is paramount—stringent regulation is something that we are very much in support of, and hope that our clients and account holders will want to have their say.
Sharon Bennett, BACN Chair, said, "The British Association of Cosmetic Nurses supports any move towards regulation which puts patient safety at the heart of its agenda. Together we will be working with BCAM, and other stakeholders to ensure that any member of the public undergoing a medical aesthetic treatment is given the 'reasonable standard of care' as expected in any medical treatment".
Catherine Fairis, the President of BCAM said, "The British College of Aesthetic Medicine (BCAM) welcomes licensing, especially if it puts patient safety at the centre of its agenda by promoting the medical model of aesthetics. We want to work collaboratively with BACN and the other main stakeholders to ensure that this consultation achieves the most effectual licencing policy for aesthetics in the UK".


 Added to basket
Added to basket

 Unapplied Changes
Unapplied Changes